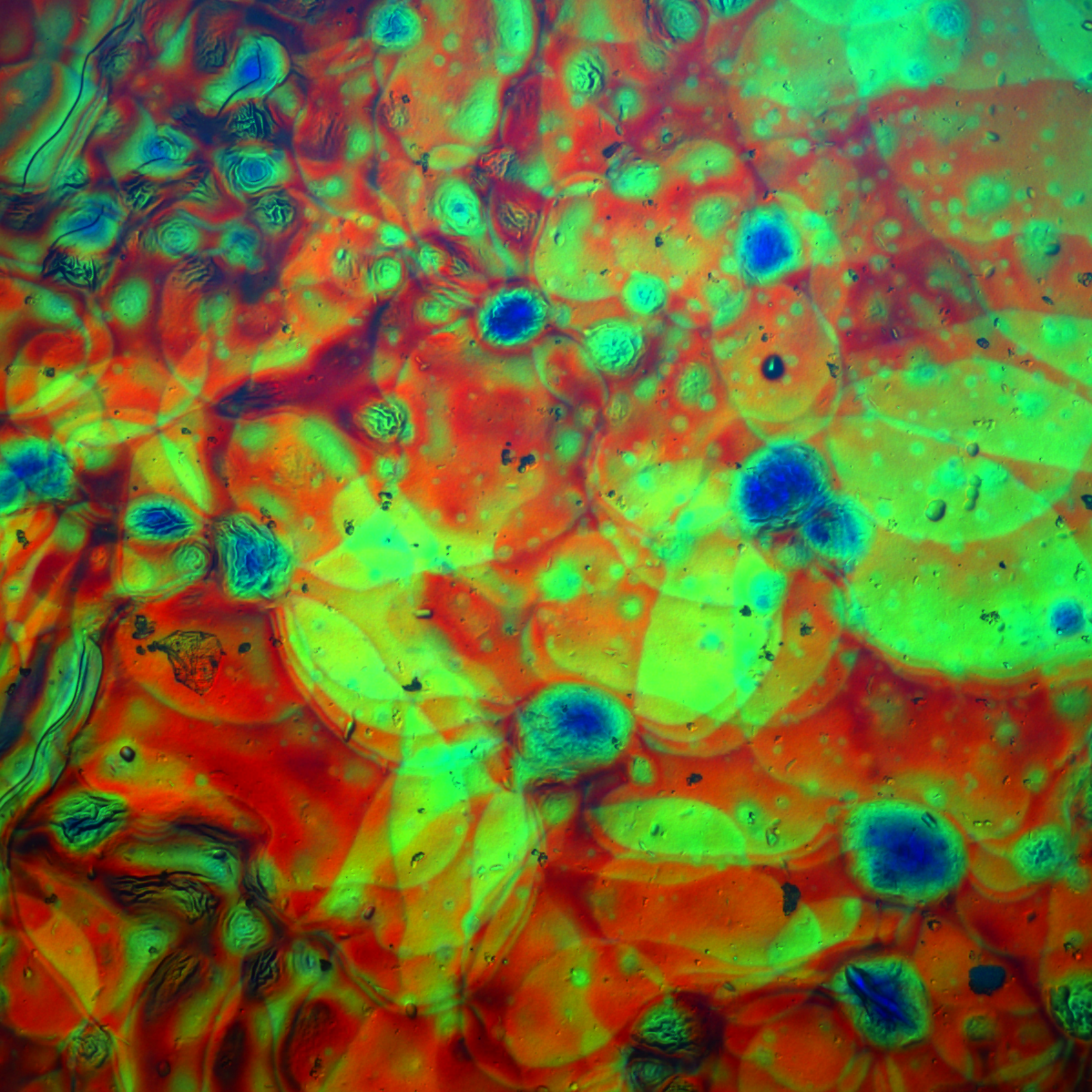
Texture of a lyotropic liquid crystal at the transition from the isotropic to the columnar phase as seen by polarizing optical microscopy.
Lyotropic Liquid Crystals
Liquid crystals make up their own state of matter, which is marked by a unique combination of fluidity and anisotropic properties, e.g. differing refractive indices, viscosities or electric polarizabilities in different directions. The key to this behaviour lies in the simultaneous presence of long-range order in some directions and its absence in other directions of the liquid crystal.
Lyotropic liquid crystals (LLCs) are a subclass of liquid crystals, which are formed by surfactant molecules, polymers or nanoparticles in combination with a solvent, typically water, under the right conditions. Various LLC phases may occur in dependence of the concentration and temperature. Investigating the structure and the stability of these LLC phases lays the foundation for most of our further work.
Transmission electron micrographs of an ordered mesoporous silica material produced by true liquid crystal templating. a) Top view of the hexagonally ordered, cylindrical pores and b) side view of the pores.
Ordered Mesoporous Materials
Mesoporous materials (pore size: 2-50 nm) possess intriguing properties and are used for a variety of applications, e.g. energy storage, synthesis of nanoparticles, adsorption or drug delivery. An elegant way to synthesize such materials is by using the self-assembled structures of surfactant-based LLCs as direct templates. An added silica precursor (or other monomer), which solely dissolves in the continuous aqueous phase, may then polycondensate around the micelles during a hydrothermal treatment step. After removing the template, an inverse of the former structure is obtained. Depending on the phase of the LLC, diverse pore types and arrangements are accessible.
Our research efforts, which are closely connected to the CRC 1333, are focused on elucidating the templating process by identifying structural correlations between the lyotropic liquid crystal, the templated material and the production parameters as well as on extending the available pore dimensions. Furthermore, we are interested in templating complex LLC structures and investigate the effects of chiral templates on the mesoporous materials.
Reflection colors of a dried cellulose nanocrystal film as seen by illumination with left-handed circular polarized light. The variations in the colors originate from slight differnces in the value or orientation of the helical pitch axis in different parts of the sample.
Polysaccharide Nanocrystals
Naturally occurring polysaccharides, such as cellulose or chitin, can be transferred into nanocrystals with a length of several hundreds and a width of a couple of nanometers by acidic hydrolysis and ultrasound treatment. In aqueous suspensions, these nanocrystals form nematic phases in which the direction of the long-range orientational order, i.e. director, of the nanocrystals precesses in orthogonal direction. The distance which is needed for a full 360° rotation of the director is called helical pitch. This chirality effect originates form the chirality centers naturally present in the polysaccharides and results in macroscopically observable characteristics such as a striped pattern between crossed polarizers, the so-called finger-print texture. When drying such nanocrystal suspension under appropriate conditions, the helical sturcture may be conserved, leading to interessting properites of the dried films, e.g. selective reflection of visible light.
In our work, we strive for extending the range of accessible polysaccharide nanocrystals and elucidating the correlations between chemical and physical structure of the nanocrystals and the properties of the LLC as well as the dried films. Furthermore, we explore the potential of the polysaccharied nanocrystal films as sensors, functional coatings and robust building materials.
Have a look at this poster for some additional information.

Johanna Bruckner
Dr.Research Group Leader